- Home
- Resource
- Explore & Learn
- A Comprehensive Guide to Toxicology in Clinical Laboratories
- Home
- IVD
- By Technology Types
- By Diseases Types
- By Product Types
- Research
- Resource
- Distributors
- Company
Toxicology, the scientific study of the adverse effects of chemicals—including drugs, environmental pollutants, industrial compounds, and naturally occurring toxins—on living organisms, plays an irreplaceable and pivotal role in modern clinical laboratories. It serves as a critical bridge between basic science and clinical practice, providing essential data that guides diagnosis, treatment, and prevention of toxin-induced illnesses. With the global rising prevalence of drug abuse, particularly the misuse of prescription medications and illicit substances, coupled with the continuous introduction of novel psychoactive substances (NPS) that emerge at an unprecedented rate, the demand for accurate, reliable, and timely toxicological analyses has never been greater in clinical settings.
These analyses are not only vital for identifying the specific toxins affecting patients but also for determining their concentrations in biological samples (such as blood, urine, and tissue), evaluating the severity of intoxication, and formulating targeted therapeutic interventions. This comprehensive guide delves deeply into the intricacies of toxicology in clinical environments, covering key aspects such as sample collection and processing protocols, analytical techniques used in toxin detection, and the interpretation of test results. Additionally, it highlights recent technological advancements that have revolutionized clinical toxicology—including the adoption of mass spectrometry and molecular diagnostic tools—and addresses the unique challenges faced by laboratory professionals, such as the rapid identification of newly emerging NPS and the need for standardized testing methods. Finally, the guide explores future directions in the field, focusing on how innovation and collaboration can further enhance the role of toxicology in improving patient outcomes and public health.
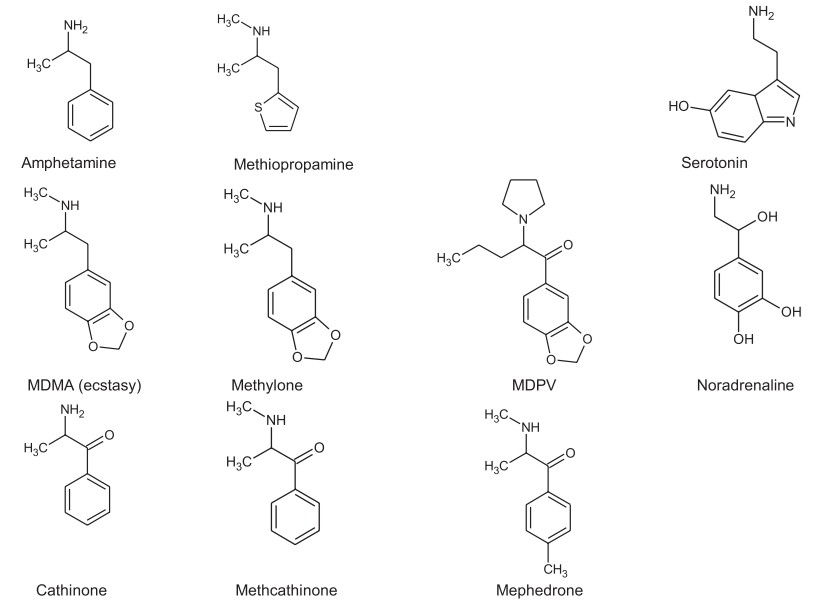 Fig.1 Structures of amphetamine, ecstasy and cathinone and a number of derivatives, with the structures of serotonin and noradrenaline for comparison. (Brown N. W., et al., 2017)
Fig.1 Structures of amphetamine, ecstasy and cathinone and a number of derivatives, with the structures of serotonin and noradrenaline for comparison. (Brown N. W., et al., 2017)
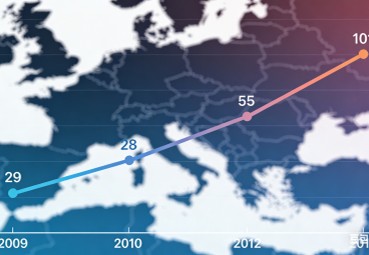
The European Monitoring Centre for Drugs and Drug Addiction (EMCDDA), the EU's leading agency dedicated to monitoring drug trends and supporting drug policy, has extensively documented a dramatic and alarming surge in the prevalence of novel psychoactive substances (NPS) across the entire European continent over the past decade. These unregulated and often dangerous substances have emerged as a major public health and safety concern, with their presence spreading rapidly across both Western and Eastern European countries, affecting communities of all ages.
In just a five-year period, the number of seized NPS reported to the EMCDDA skyrocketed from a relatively low 29 distinct substances in 2009 to an unprecedented peak of 101 different NPS in 2014—a more than threefold increase that underscored the illicit market's ability to rapidly adapt and proliferate new compounds. This sharp upturn was not limited to seizures alone; reports of NPS-related hospitalizations, overdoses, and even fatalities also rose in tandem, further highlighting the urgent need for improved detection and response measures.
These substances, often deceptively marketed as "legal highs," "herbal blends," or "research chemicals" to evade regulatory restrictions, have introduced a new and complex layer of challenges for toxicologists working in clinical, forensic, and public health settings. Unlike traditional illicit drugs (such as heroin, cocaine, or cannabis) with well-documented chemical properties, NPS encompass a vast and diverse range of chemical structures, many of which are modified versions of existing psychoactive compounds to bypass legal controls.

Detecting NPS in clinical samples is further complicated by their extensive metabolism. Many of these substances undergo significant biotransformation, leading to low or undetectable levels of the parent compound in urine. This metabolic complexity means that traditional screening methods, such as immunoassays, are often insufficient for detecting NPS. Instead, advanced mass spectrometric techniques, like liquid chromatography-tandem mass spectrometry (LC-MS/MS) and high-resolution mass spectrometry (HRMS), are increasingly relied upon to identify these substances.
For example, synthetic cannabinoids (SC), a class of NPS that bind to cannabinoid receptors with high affinity, are frequently detected in products marketed as "herbal" or "spice" blends. These compounds undergo complex metabolism, producing numerous metabolites that may be more detectable than the parent drug. However, the lack of standardized reference materials and the constant emergence of new SC variants make identification a daunting task. Each new variant may have a slightly different chemical structure, requiring updated detection methods and reference standards. This rapid evolution of NPS underscores the need for flexible and adaptable analytical techniques that can keep up with the changing landscape of drug use.

Rapid Screening with Immunoassays
Immunoassays, particularly automated enzyme-linked immunosorbent assays (ELISAs), are widely used in clinical laboratories for rapid drug screening. These assays offer high throughput, ease of use, and relatively low cost, making them ideal for initial screening in various healthcare settings. Many instant point-of-care tests (POCTs) also utilize immunoassay technology, enabling on-site drug testing in emergency departments, primary care clinics, and even in the field. The ability to provide quick results allows healthcare providers to make timely decisions regarding patient care, ensuring that individuals receive appropriate interventions as soon as possible.
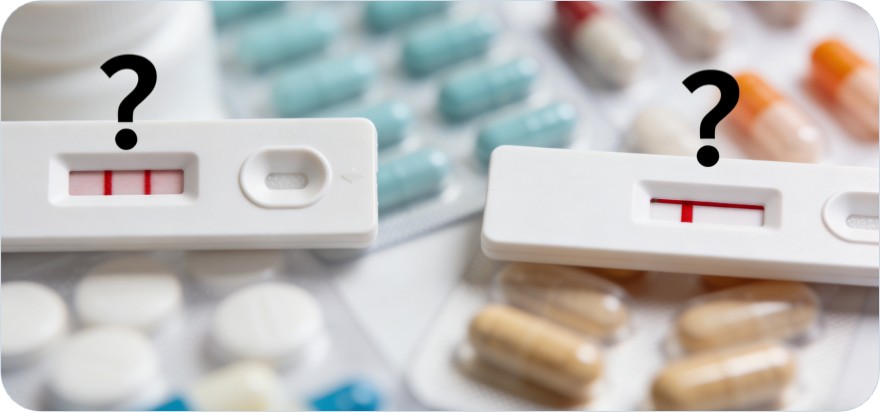
False Positives and Negatives
Despite their widespread use and numerous advantages, immunoassays are prone to false positives and negatives, which can significantly impact the accuracy of drug screening results. Cross-reactivity with structurally similar compounds is a common issue that can lead to false positive results. For example, certain medications and dietary supplements have been reported to cause false positives in amphetamine and benzodiazepine immunoassays. Conversely, low drug concentrations or the presence of masking agents may result in false negatives, potentially overlooking substance use when it is present. These inaccuracies highlight the importance of using confirmatory testing methods, such as mass spectrometry, to validate initial screening results and ensure the reliability of drug testing outcomes.
If you have related needs, please feel free to contact us for more information or product support.
Reference
This article is for research use only. Do not use in any diagnostic or therapeutic application.
|
There is no product in your cart. |